A Standard Guide for Laboratory Information Management Systems (LIMS)
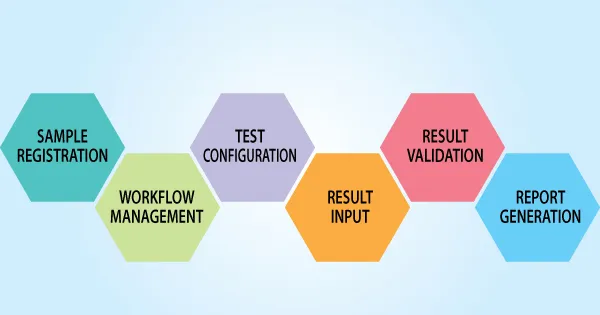
In the dynamic landscape of modern laboratories, where the management of vast datasets and regulatory compliance is paramount, Laboratory Information Management Systems (LIMS) emerge as indispensable tools. To ensure the effective implementation and utilization of LIMS, a standardized guide becomes essential. This article aims to delve into the key aspects of the baytek standard guide for Laboratory Information Management Systems, providing laboratories with a roadmap for successful adoption and utilization.
The Significance of Standardization in LIMS:
Ensuring Consistency Across Laboratories: A standardized guide for LIMS sets the foundation for consistency across different laboratories. By adhering to a common set of practices and principles, laboratories can achieve uniformity in data management, reporting, and regulatory compliance.
Facilitating Interoperability: Standardization enables interoperability, allowing different LIMS to communicate seamlessly with other laboratory instruments and systems. This ensures that data flows smoothly across various platforms, promoting efficiency and reducing the risk of errors.
Enhancing Regulatory Compliance: Compliance with industry regulations is a critical aspect of laboratory operations. A standardized guide for LIMS provides a framework for meeting regulatory requirements, helping laboratories navigate complex standards and ensuring data integrity and security.
Key Components of the Standard Guide for LIMS:
Requirements Analysis: The first step in the standard guide involves a comprehensive analysis of the laboratory’s requirements. This includes understanding the laboratory’s size, type of analysis performed, and specific workflow needs. The guide helps laboratories tailor their LIMS to meet their unique requirements.
Vendor Selection Criteria: Choosing the right LIMS vendor is crucial for successful implementation. The standard guide outlines criteria for vendor selection, considering factors such as the vendor’s reputation, track record, customer support, and the flexibility of the LIMS solution to accommodate future growth.
Installation and Configuration: The guide provides a roadmap for the installation and configuration of the LIMS. It covers aspects such as hardware requirements, network considerations, and database configurations. Clear guidelines ensure a smooth implementation process and minimize the risk of technical challenges.
User Training and Adoption: Successful adoption of LIMS relies heavily on user training. The standard guide includes recommendations for developing training programs, documentation, and ongoing support strategies. Ensuring that laboratory staff is well-versed in using the system optimally is key to maximizing the benefits of LIMS.
Data Migration Strategies: Migrating existing data to the new LIMS is a critical cbliss phase. The guide offers strategies for efficient data migration, minimizing downtime and potential data loss. It includes considerations for mapping data fields, validating migrated data, and ensuring data accuracy during the transition.
Customization and Configuration: Laboratories have diverse workflows, and a one-size-fits-all approach may not be suitable. The guide provides insights into customization and configuration options, allowing laboratories to adapt the LIMS to their specific needs without compromising standardization.
Quality Assurance and Control: Maintaining data quality and ensuring accurate results are central to laboratory operations. The standard guide outlines quality assurance and control measures within the LIMS, covering aspects such as audit trails, data validation, and the implementation of quality management systems.
Security Protocols: Data security is a paramount concern in laboratory environments, especially when dealing with sensitive information. The guide addresses security protocols, including user access controls, encryption, and regular security audits, to safeguard data against unauthorized access and cyber threats.
Maintenance and Upgrades: The lifecycle of a LIMS involves ongoing maintenance and periodic upgrades. The standard guide offers recommendations for establishing maintenance schedules, conducting regular system audits, and managing software updates to ensure that the LIMS remains current and secure.
Documentation Standards: Clear and comprehensive documentation is crucial for the continued success of LIMS implementation. The guide provides standards for documentation, including user manuals, system architecture diagrams, and change management procedures.
Benefits of Adhering to the Standard Guide:
Operational Efficiency: Following a standardized guide streamlines the implementation process, reducing the likelihood of errors and minimizing downtime. This, in turn, enhances overall laboratory efficiency and productivity.
Data Integrity and Compliance: Adherence to a standard guide ensures that the LIMS meets regulatory requirements and maintains data integrity. Laboratories can confidently demonstrate compliance during audits, reducing the risk of penalties and legal complications.
Cost-Effectiveness: A standardized approach to LIMS implementation contributes to cost-effectiveness. By avoiding costly errors and rework, laboratories can optimize their investments in LIMS and achieve long-term financial benefits.
Scalability and Adaptability: The standard guide provides a foundation for scalable and adaptable LIMS solutions. Laboratories can confidently expand their operations or adapt to changing needs without the need for extensive system overhauls.
Interoperability with Other Systems: Standardization promotes interoperability, allowing LIMS to seamlessly integrate with other laboratory instruments and systems. This interconnectedness enhances data flow and collaboration across different platforms.
Conclusion:
The standard guide for Laboratory Information Management Systems serves as a compass for laboratories navigating the complexities of LIMS adoption. By providing a systematic approach to requirements analysis, vendor selection, installation, training, and ongoing maintenance, the guide empowers laboratories to optimize their use of LIMS. Standardization not only ensures consistency and compliance but also contributes to operational efficiency, cost-effectiveness, and adaptability. As laboratories continue to evolve, a standardized guide for LIMS remains an invaluable resource, guiding them toward excellence in data management and laboratory operations.